Chapter 3 Practice Problems
Answers for these practice problems are on the next page.
A good approach is to answer all of the questions on a piece of paper and then check your answers. This avoids accidentally seeing the answer(s) for questions you have not done yet.
Q3.1: Draw and label Newman Projections for the antiperiplanar, fully eclipsed, both partially eclipsed, and both gauche conformations of 1-iodopropane. For simplicity, use a perspective that has the iodine closer to the viewer in each case.
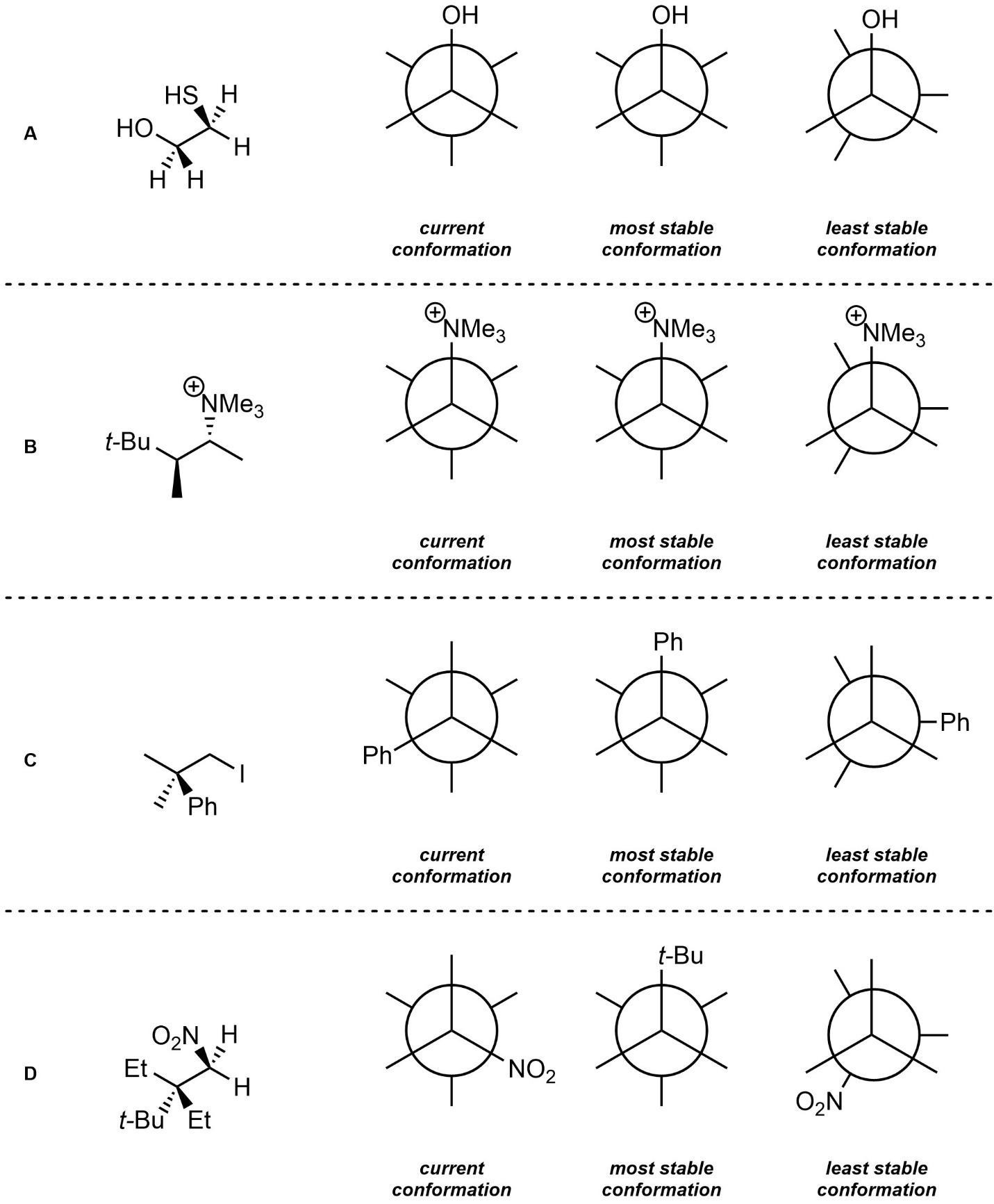
Q3.2: For each of the following molecules add the remaining substituents to complete Newman Projections of the current, most stable, and least stable conformations. It is not necessary to draw out full groups; using the abbreviations provided is acceptable. However, knowing what the abbreviations represent will assist in answering the question.
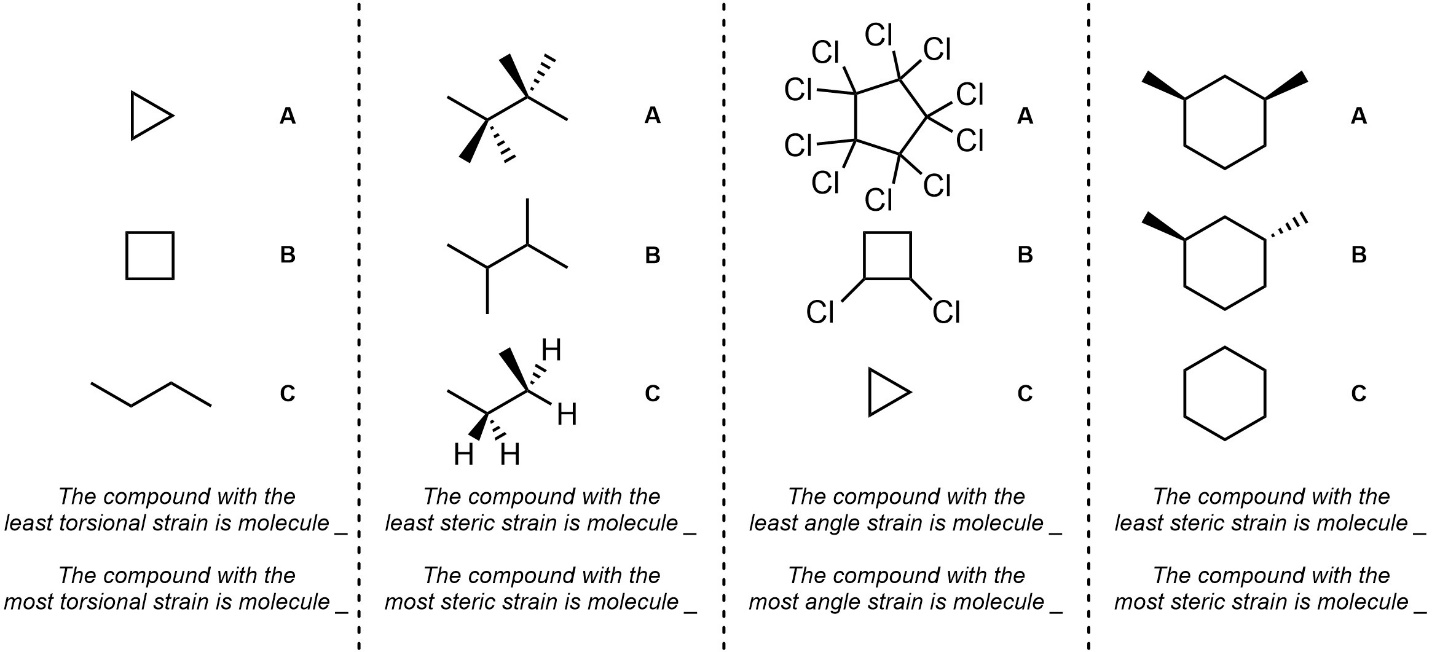
Q3.3: Assume each of the following adopts the lowest energy conformation available to it. Fill in the blanks for each set of molecules by assessing the strains they experience.
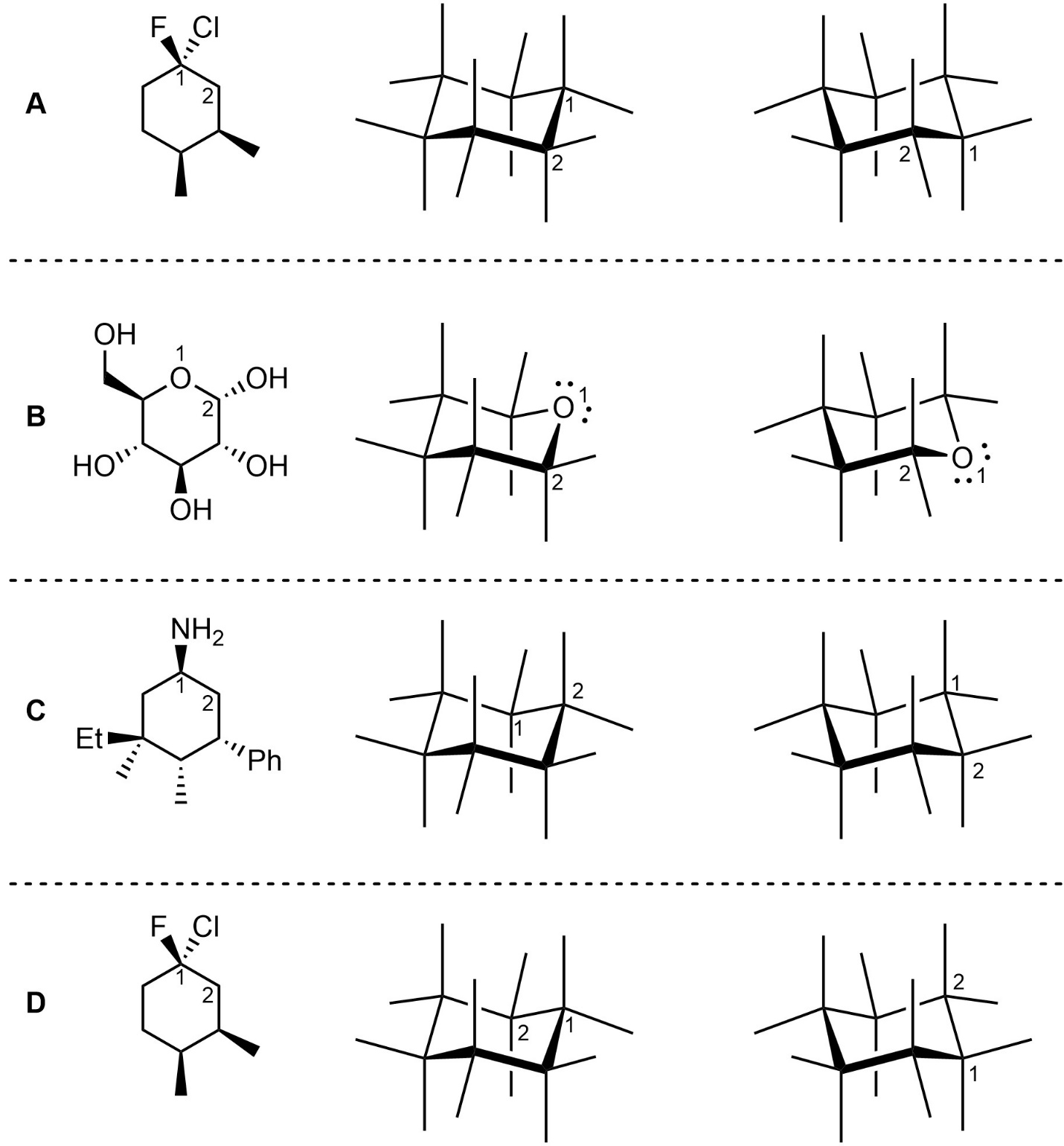
Q3.4: For each six-membered ring draw both chair conformations and then label (highlight, colour-code, etc.) all substituents other than H’s as axial or equatorial. A template of each chair is provided to set the perspective. The perspective outlined must be used. For practice purposes you should attempt to draw these chair conformations (in the same perspective) by hand.
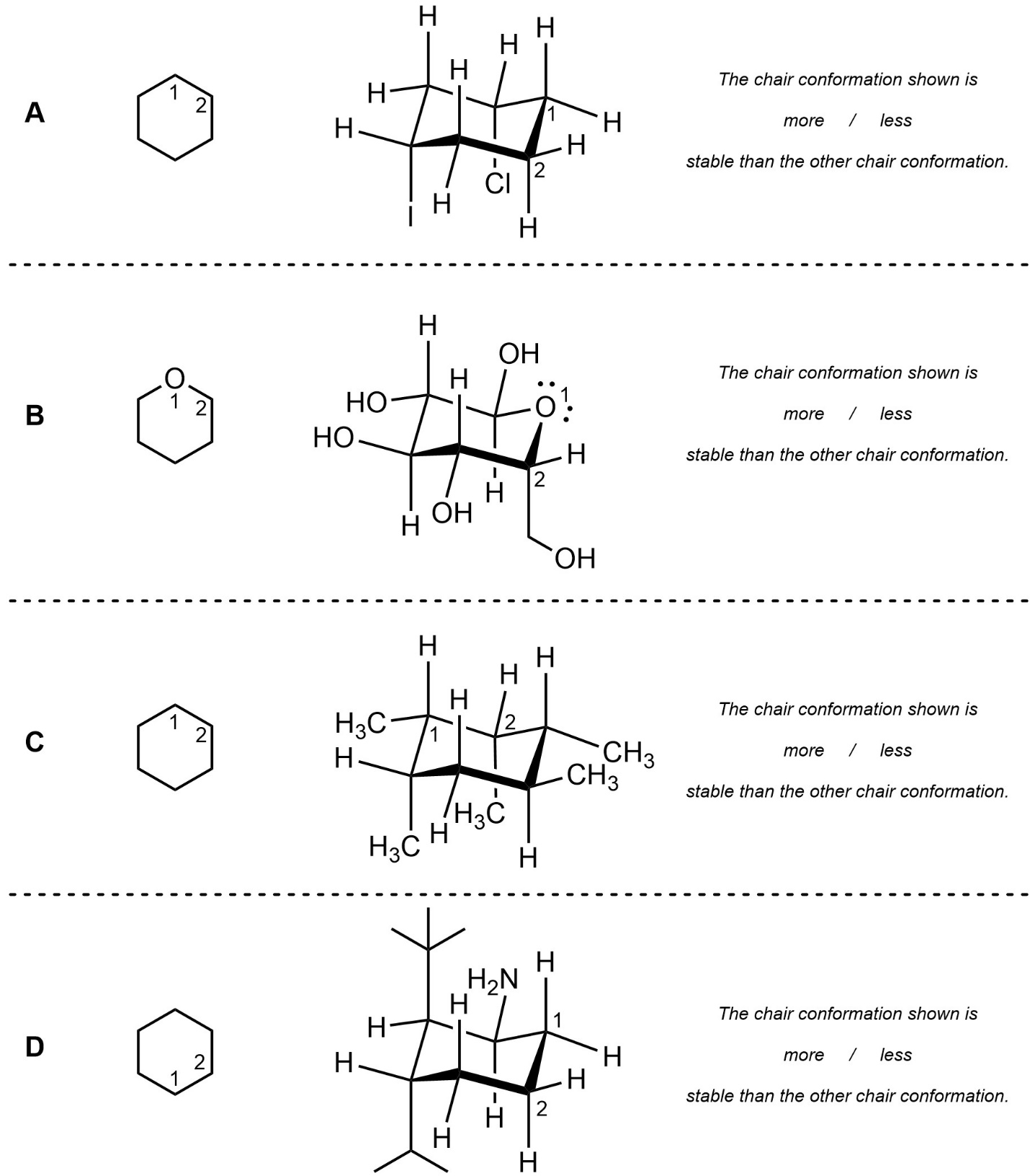
Q3.5: For each chair conformation below add the substituents to the line-angle structure (including hashed/wedged notation) and indicate whether the chair conformation shown is more or less stable than the other chair conformation. You do not need to draw the other chair conformation, though doing so may assist in answering the question.