1.7. Hashed and Wedged Notation
More advanced ways of depicting molecules (see Section 1.8) attempt to show three-dimensional structure in their construction. There are a variety of reasons this is useful, but the primary reason involves the accurate portrayal of stereoisomers (see Chapter 4).
The most common notation for showing three-dimensional orientation is the hashed/wedged system. In this approach simple lines are used to represent bonds that occupy the plane of the page (screen, board, etc.). Hashed bonds are drawn to indicate that the attached atom is oriented away from the viewer (i.e. behind the plane of the page). Wedged bonds are drawn to indicate that the attached atom is oriented towards the viewer (i.e. in front of the plane of the page). This allows us to show three-dimensional geometries, such as the tetrahedral geometry of methane (Figure 1.36).
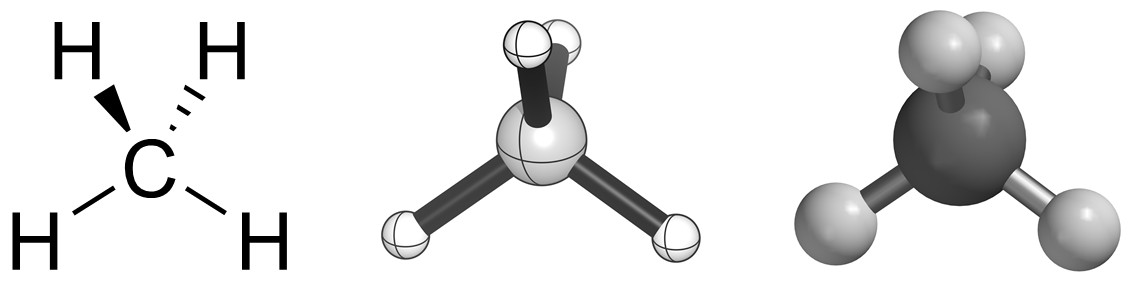
Figure 1.36 – Three Representations of Methane Using Hashed/Wedged Bonds and Ball-and-Stick Models.
To be drawn properly the hashed/wedged bonds should start narrow at the atom that lies in the plane of the page and widen as they approach the atom behind/in front of the plane. Some older depictions lack this (Figure 1.37). This is fine for simple molecules but can be confusing or ambiguous in complicated molecules. As such, it should be avoided.
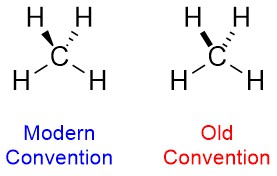
Figure 1.37 – Modern vs. Old Conventions for Hashed/Wedged Bonds.
To be drawn properly the hashed/wedged bonds should both be on the same side and should be made to resemble a tetrahedron (Figure 1.38).
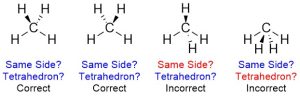
Figure 1.38 – Accurately Portraying Three-Dimensional Geometry Using Hashed/Wedged Bonds.
The left-to-right order of hashed vs. wedged bonds is not important; the hashed/wedged system is an imperfect way of indicating the two groups are directly in front of each other from the viewer’s perspective (Figure 1.39).
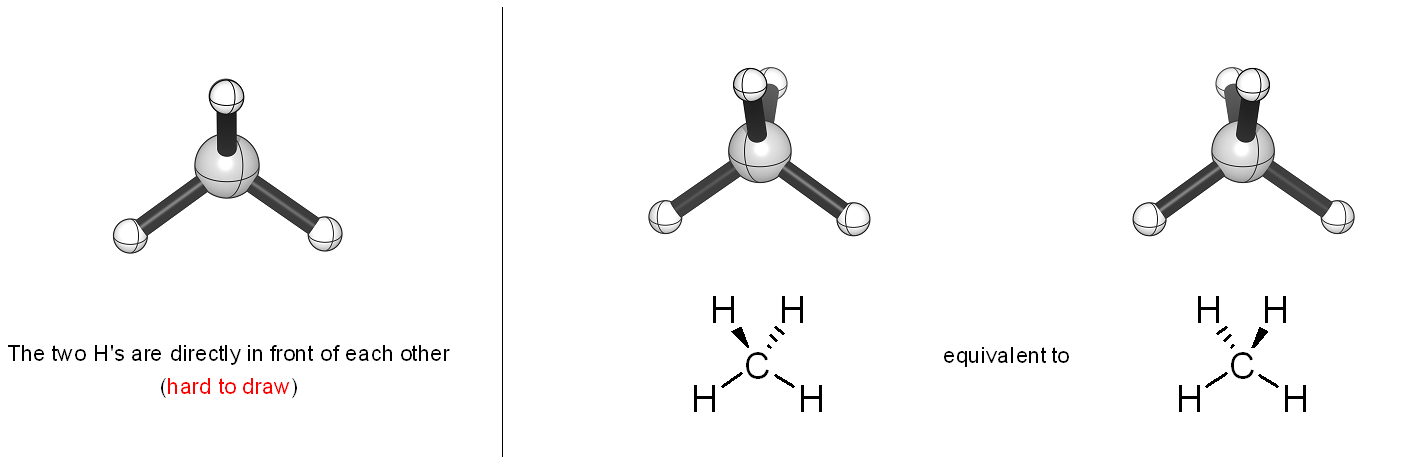
Figure 1.39 – Left-to-Right Order for Drawing Hashed and Wedged Bonds.

