Chapter 2 Practice Problems – Answers
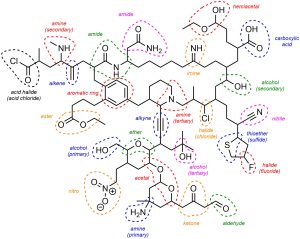
Q2.1: The following hypothetical molecule contains an impressive 25 functional groups. One of them has been circled and named for you. Circle and name the remaining functional groups in the same fashion. If possible, describe the subdivision (e.g. primary/secondary/tertiary, etc.). Functional groups may appear more than once. You may ignore alkanes.
Colour added for clarity (not required).
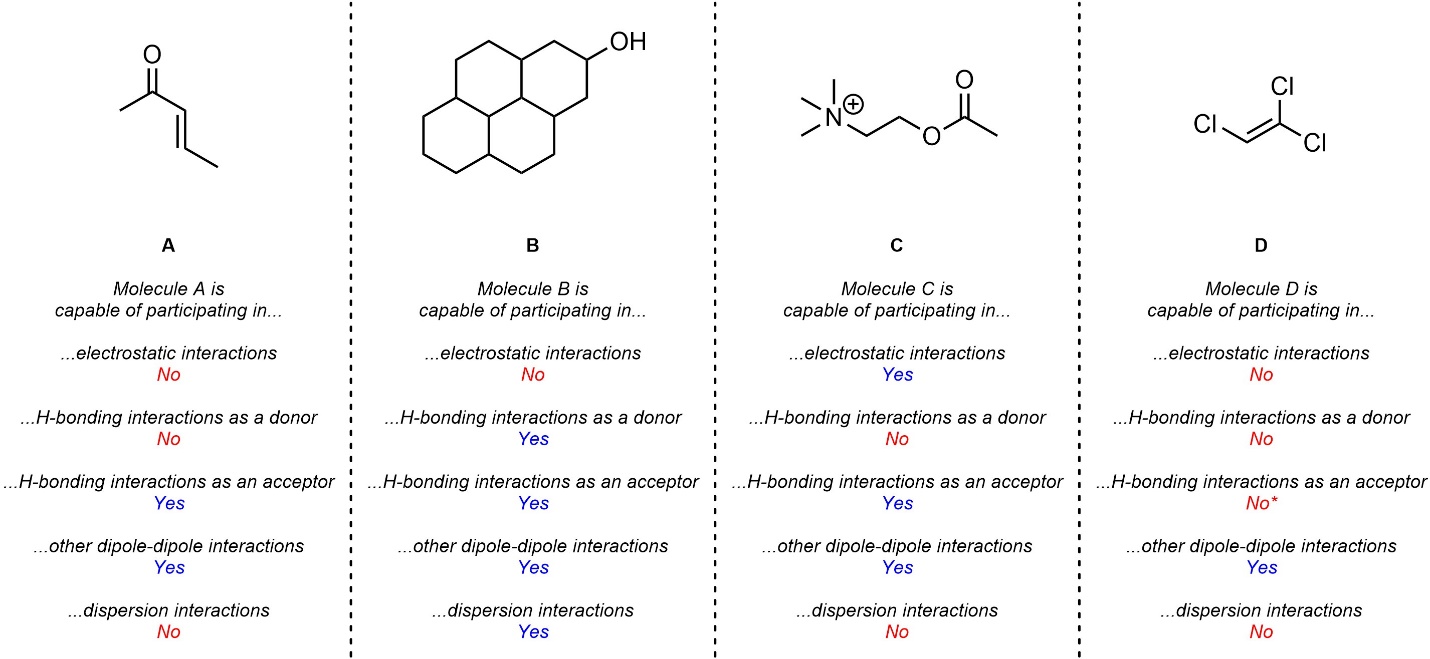
Q2.2: Which intermolecular force(s) is/are each of the following molecules capable of? For simplicity, only consider dispersion if it is the only force present or there is a large area that could interact via it alone.
Revisiting the definitions of H-bonding donor/acceptor will help.
NOTES:
Technically all molecules are capable of and interact via dispersion.
However, the effect is normally small enough that
it is overshadowed by (any) other intermolecular force(s).
Theoretically Cl can act as a H-bond acceptor. In practice this works poorly.
Q2.3: Below are three solvents. For each molecule choose which of the three solvents would be best for maximizing solubility.
Classifying Polar/Non-Polar // Protic/Aprotic will help.
One important exception to consider…
Salts are VERY water soluble.
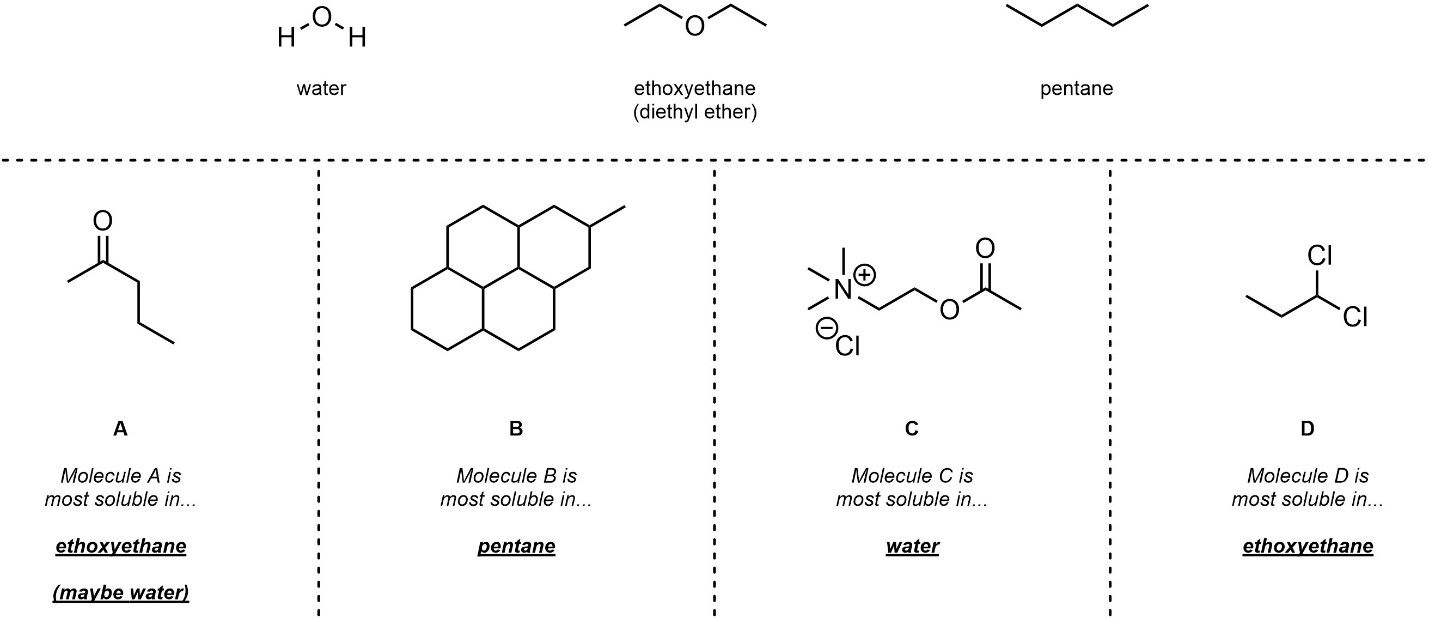
Electrostatic interactions make molecules VERY water soluble.
(usually overrides all other factors)
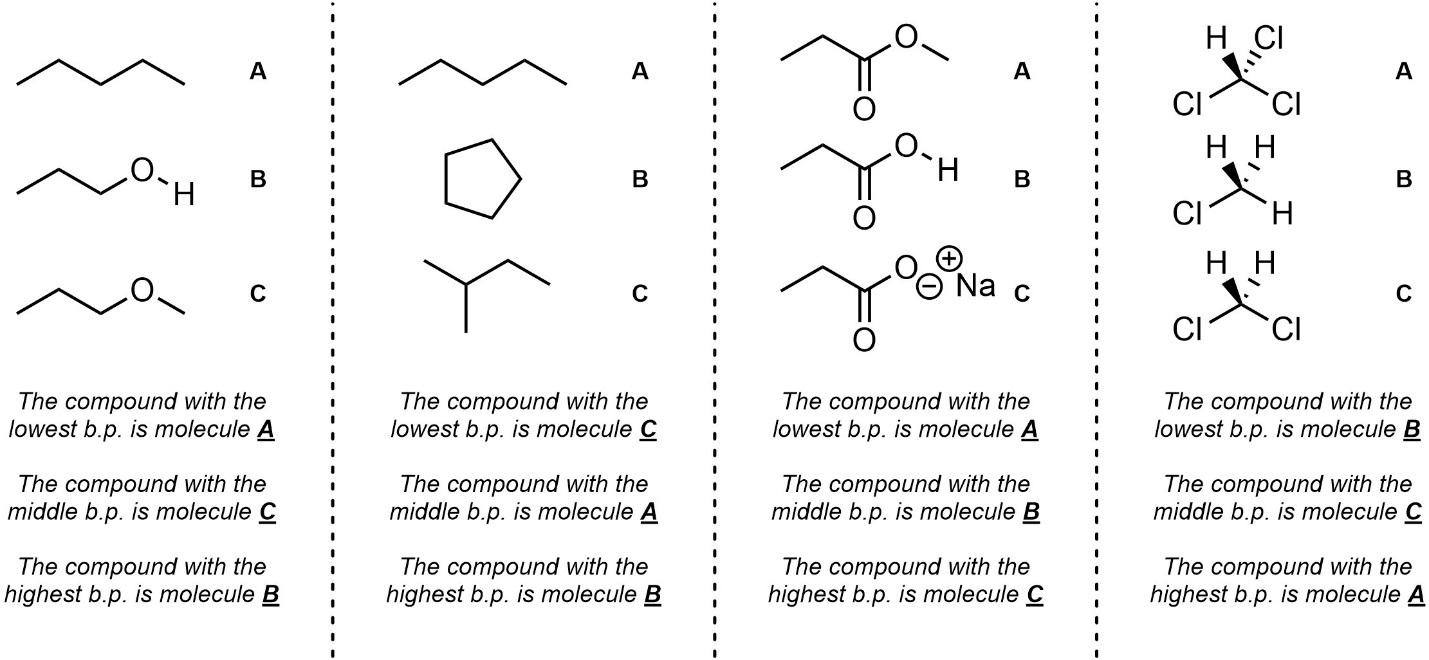
Q2.4: Rank each set of molecules in order of increasing boiling point (lowest to highest).
Q2.5: Name each of the following molecules using the IUPAC rules discussed in the text. Be aware that names may be (significantly) longer/shorter than the blank spaces suggest.
Use the tables.
(priority, suffix/prefix, chain lengths, Greek descriptors, trivial names, etc.)
The method in Section 2.5.9 may help.
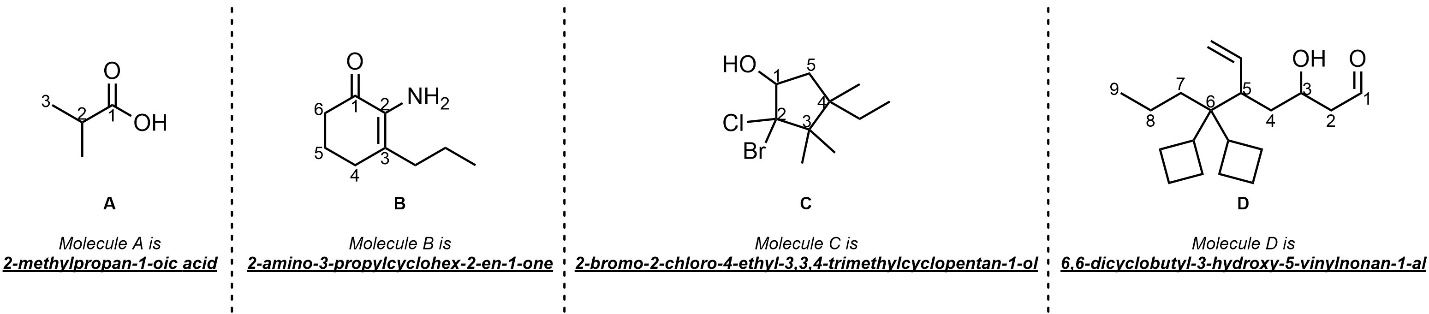
These examples are meant to be semi-challenging and time-consuming
for practice purposes.
Q2.6: Draw each molecule using the provided IUPAC name.
Use the tables.
(priority, suffix/prefix, chain lengths, Greek descriptors, trivial names, etc.)
The method in Section 2.5.10 may help.
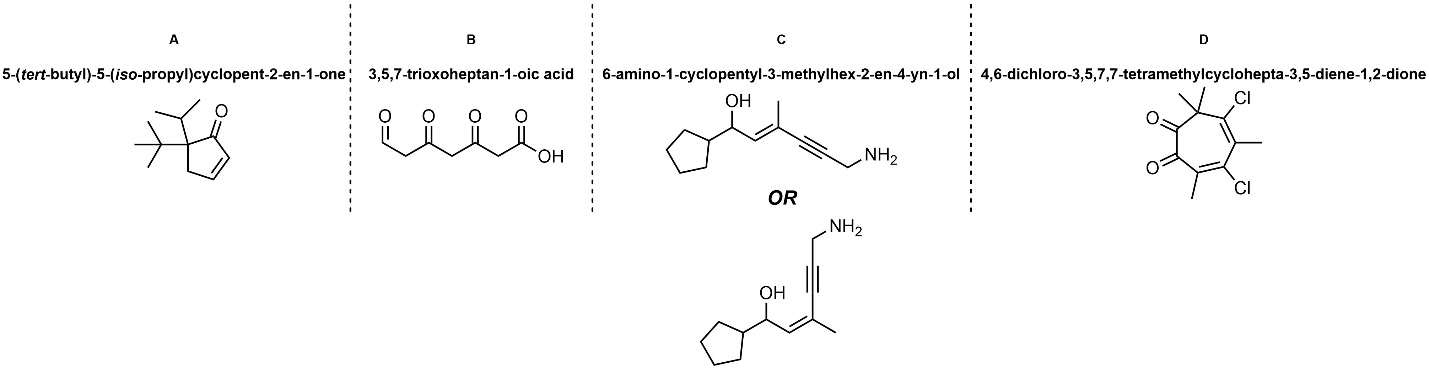
These examples are meant to be semi-challenging and time-consuming
for practice purposes.

