10.8. Reaction: Alkylation via Friedel-Crafts
It is possible to change a hydrogen (H) into an alkyl group on an aromatic ring (Scheme 10.10). There are multiple ways of accomplishing this. The most common way involves the use of an alkyl halide and a Lewis acid catalyst. This is often referred to as Friedel-Crafts Alkylation in honour of the developers of this reaction.
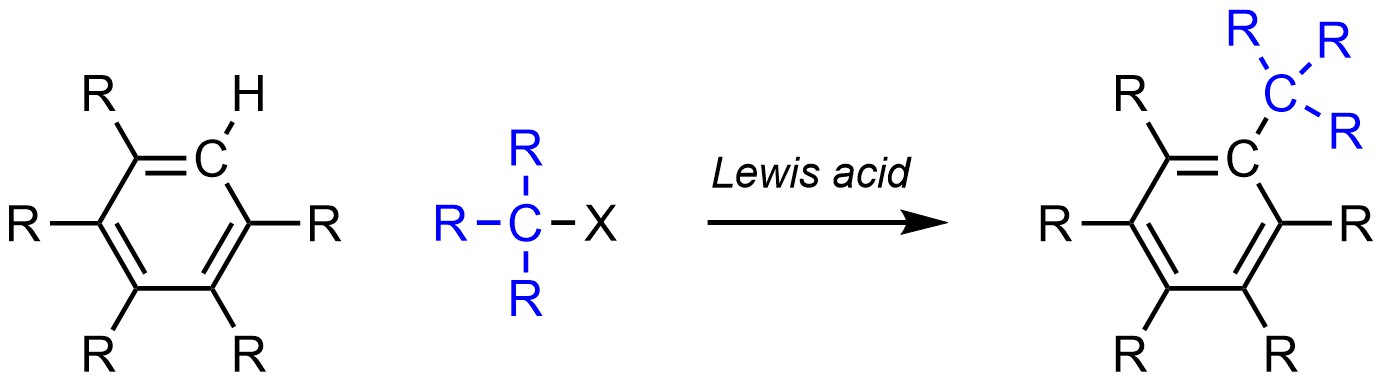
Scheme 10.10 – Generalized Reaction Equation for Friedel-Crafts Alkylation of an Aromatic Ring.
10.8.1. Catalyst Requirements
Directly using an alkyl halide will not result in alkylation without a catalyst. The catalysts used are Lewis acids, and work by coordination with the halide. Although many Lewis acids are able to catalyze these reactions (see Table 10.1), FeBr3 and AlCl3 are the most commonly used. This is due to convenience rather than any catalytic benefit over other Lewis acids.
10.8.2. Mechanism
The overall mechanism for this reaction has the same two main steps but has an additional activation step for the catalyst (Scheme 10.11). The alkyl halide (nucleophile) reacts with the Lewis acid catalyst (electrophile) to form an ionic complex. This greatly increases its electrophilicity. Depending on the exact structure of the alkyl group, the ionic complex may or may not form a carbocation. Then, a π bond from the aromatic ring (nucleophile) attacks the carbon (activated electrophile). This creates a new C-C bond and a carbocation. Finally, the halide (nucleophile/base) removes the original hydrogen (electrophile/acid), which regenerates aromaticity and the catalyst. Although halides are very weak bases, the last step is favourable because it regenerates aromaticity.
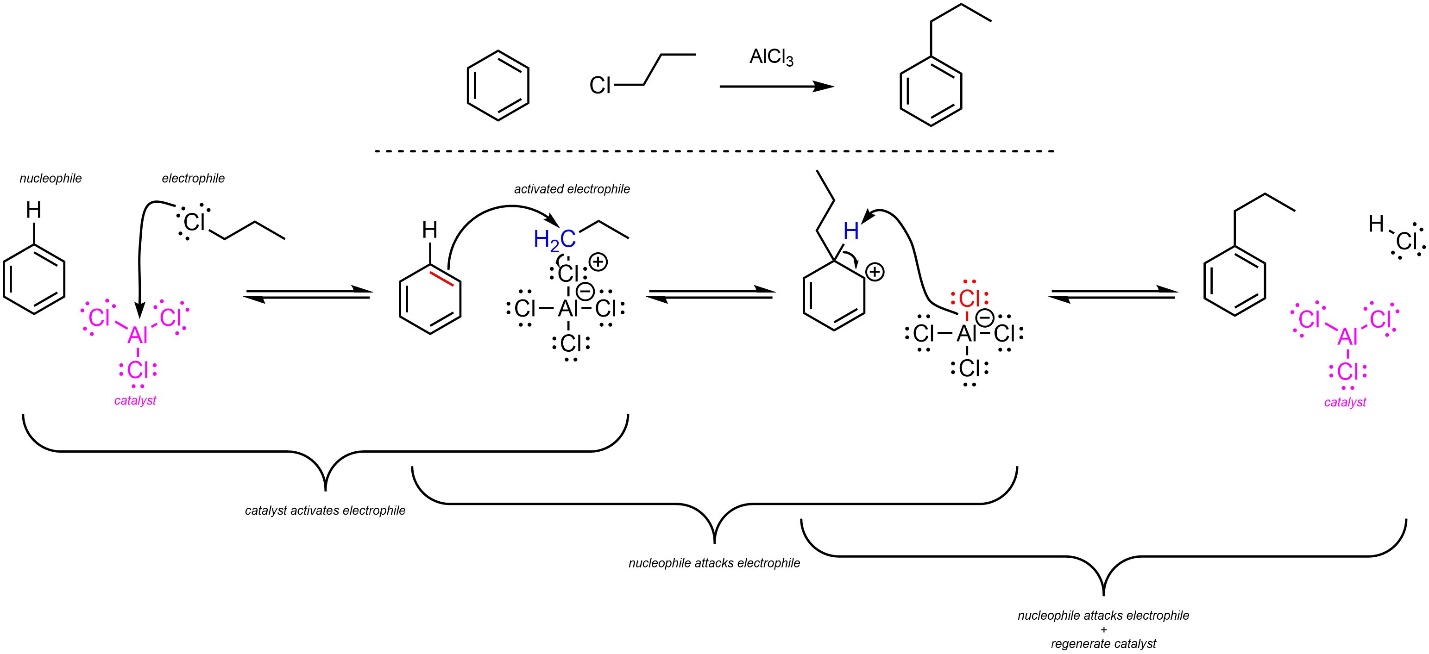
Scheme 10.11 – Reaction Mechanism for Friedel-Crafts Alkylation of Benzene Using 1-Chloropropane and Aluminum Trichloride.
10.8.3. Limitations and Side Reactions
Friedel-Crafts alkylation will not work well if the aromatic ring possesses any functional group that has one or more lone pairs (Figure 10.2). Functional groups with lone pairs will react with the Lewis acid catalyst.
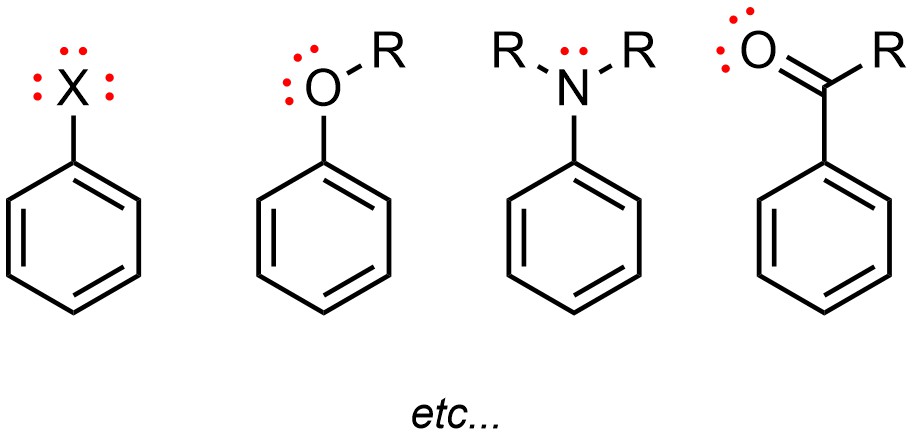
Figure 10.2 – Examples of Aromatic Rings Which Cannot Undergo Friedel-Crafts Alkylation.
Friedel-Crafts alkylation has two additional challenges.
First, it is often difficult to only alkylate the aromatic ring a single time (Scheme 10.12). After the first alkylation the product is more reactive than the starting material (see Section 10.10.1.), so it may undergo additional alkylation(s). This often results in low yield of the desired product.
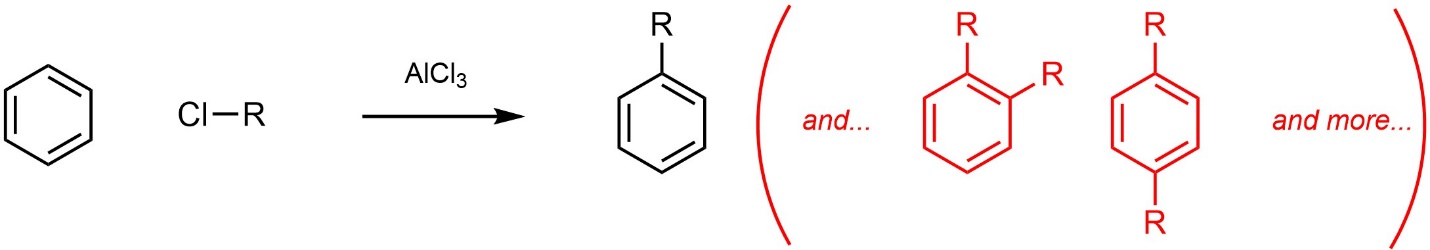
Scheme 10.12 – Examples of Multiple Alkylation Side Products in Friedel-Crafts Alkylation.
Second, depending on the exact structure of the alkyl halide it may form a carbocation intermediate (Scheme 10.13). Some carbocations, particularly primary carbocations without resonance stabilization, may undergo other reactions instead of (or in addition to) the desired reaction. The specifics of these other reactions lie outside the scope of this text. This often results in low yield of the desired product.
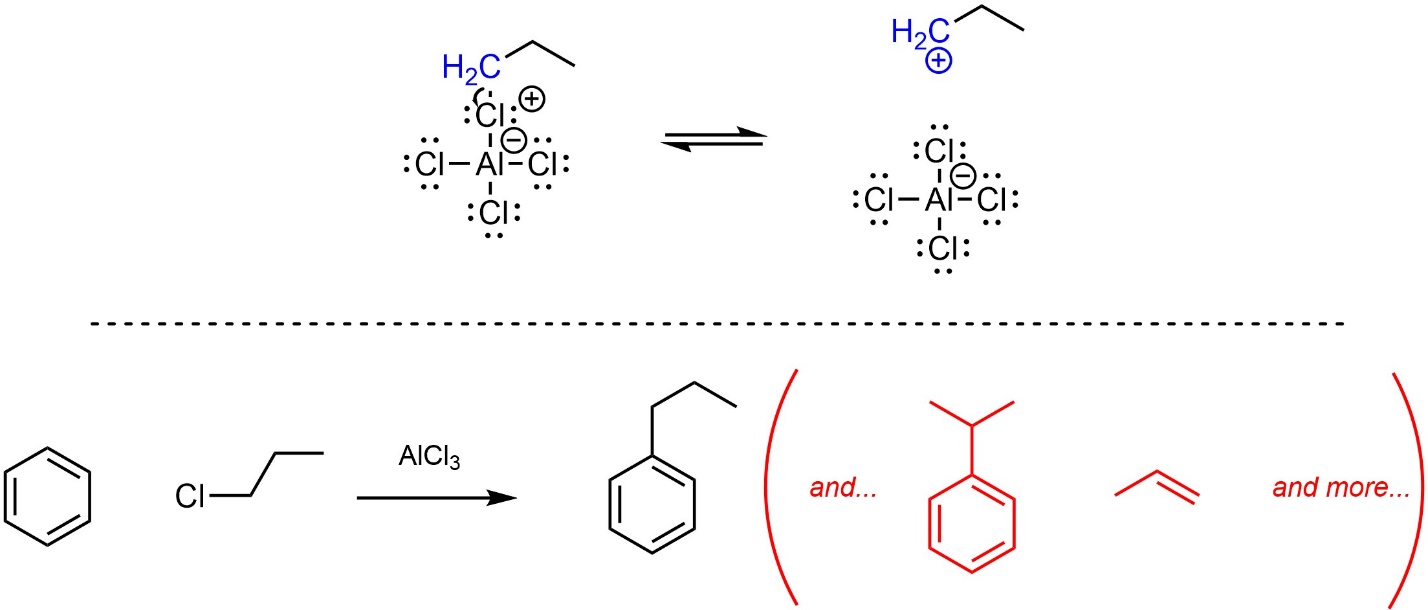
Scheme 10.13 – Examples of Other Side Products in Friedel-Crafts Alkylation.
At an introductory level it is difficult to predict whether a given Friedel-Crafts alkylation reaction will experience these issues. This text will specify when students should consider the possibility of other products being formed during Friedel-Crafts alkylation reactions. In all other cases, assume the reaction will work as desired.

