5.6 Mineral Properties
Minerals are universal. A crystal of hematite on Mars will have the same properties as one on Earth, and the same as one on a planet orbiting another star. That’s good news for geology students who are planning interplanetary travel, because they can use the same properties to identify minerals anywhere. That doesn’t mean that it’s easy, however. Identification of minerals takes practice. Some of the mineral properties that are useful for identification are colour, streak, lustre, hardness, habit, cleavage or fracture, and density.
Colour
Some minerals have distinctive colours that useful as diagnostic criteria. The mineral sulphur (Figure 5.30 left) is always a characteristic bright yellow. For other minerals, colour might vary. Hematite is an example of a mineral for which colour is not necessarily diagnostic. In some forms hematite is a deep dull red (a fairly unique colour), but in others it is a metallic silvery black (5.30, right).

For other minerals, the problem is that a single mineral can have a wide range of colours. The colour variations can be the result of varying proportions of trace elements within the mineral, or structural defects within the crystal lattice. In the case of quartz (Figure 5.31), milky quartz gets its white colour from millions of tiny fluid-filled cavities. Smoky quartz gets its grey colour from structural damage caused by natural radiation. Amethyst and citrine get their colours from trace amounts of iron, and rose quartz gets its pink hue from manganese.

Streak
The colour of a mineral is what you see when light reflects off the surface of the sample. One reason that colour can be so variable is that the surface textureis variable. A way to get around this problem is to grind a small amount of the sample to a powder and observe the colour of the powder. This colour is the mineral’s streak. The mineral can be powdered by scraping the sample across a piece of unglazed porcelain called a streak plate (Figure 5.32). In Figure 5.32, two samples of hematite have been scraped across the streak plate. Even though one sample is metallic and the other is deep red, both have a similar reddish-brown streak.

Streak is an especially helpful property when minerals look similar. In Figure 5.33 all of the minerals are dark in colour, with varying degrees of metallic sheen. The streaks of these minerals are much more distinctive.

Lustre
Lustre is the way light reflects off the surface of a mineral, and the degree to which it penetrates into the interior. The key distinction is between metallic and non-metallic lustre. Light does not pass through metals, and that is the main reason they look metallic (e.g., the hematite on left of Figure 5.32). Even a thin sheet of metal — such as aluminum foil — will be not permit light to pass through it. Many non-metallic minerals may look as if light will not pass through them, but if you look closely at a thin slice of the mineral you will see that the mineral is translucent or transparent.
If a non-metallic mineral has a shiny, reflective surface, it is said to have a glassy lustre. The quartz crystals in Figure 5.31 are examples of minerals with glassy lustre. If the mineral surface is dull and non-reflective, it has an earthy lustre (like the hematite on the right of Figure 5.32). Other types of non-metallic lustres are silky, pearly, and resinous. Lustre is a good diagnostic property because most minerals will always appear either metallic or non-metallic, although as Figure 5.31 shows, there are exceptions.
Hardness
One of the most important diagnostic properties of a mineral is its hardness. In practical terms, hardness determines whether or not a mineral can be scratched by a particular material.
In 1812 German mineralogist Friedrich Mohs came up with a list of 10 minerals representing a wide range of hardness, and numbered them 1 through 10 in order of increasing hardness (Figure 5.34, horizontal axis). While each mineral on the list is harder than the one before it, the measured hardness (vertical axis) is not linear. Notice that apatite is about three times harder than fluorite, and diamond is three times harder than corundum.
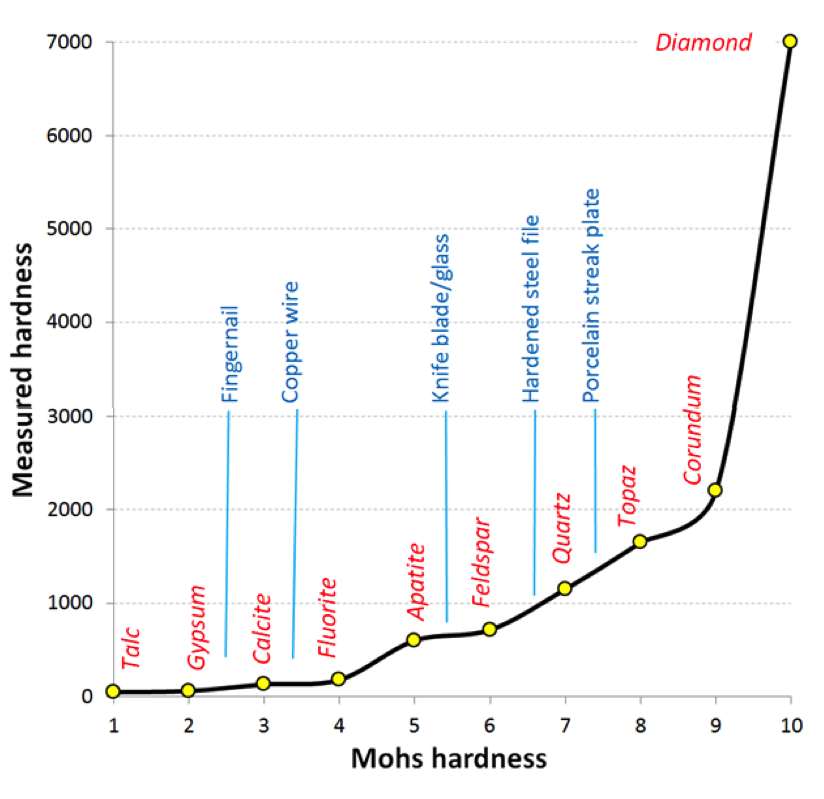
Some commonly available reference materials are also shown on this diagram, including a typical fingernail[1] (2.5), a piece of copper wire (3.5), a knife blade or piece of window glass (5.5), a hardened steel file (6.5), and a porcelain streak plate (7). These are tools that a geologist can use to measure the hardness of unknown minerals: if you have a mineral that you can’t scratch with your fingernail, but you can scratch with a copper wire, then its hardness is between 2.5 and 3.5. The minerals themselves can be used to test other minerals.
Crystal Habit
When minerals form within rocks, there is a possibility that they will form in distinctive crystal shapes if they are not crowded out by other pre-existing minerals. Every mineral has one or more distinctive crystal habits determined by their atomic structure, although it is not that common in ordinary rocks for the shapes to be obvious.
Quartz, for instance, will form six-sided prisms with pointed ends (Figure 5.35 left), but this typically happens only when it crystallizes from a hot water solution within a cavity in an existing rock. Pyrite can form cubic crystals (Figure 5.35 centre), but can also form crystals with 12 faces, known as dodecahedra. The mineral garnet also forms many-sided crystals with an over-all rounded shape (Figure 5.35 right).

Some of the terms that are used to describe habit include bladed, botryoidal (grape-like), dendritic (branched), drusy (an encrustation of crystals), equant (similar size in all dimensions), fibrous, platy, prismatic (long and thin), and stubby.
Cleavage and Fracture
Cleavage and fracture describe how a mineral breaks. These characteristics are the most important diagnostic features of many minerals, and often the most difficult to understand and identify. Cleavage is what we see when a mineral breaks along a plane or planes, while fracture is an irregular break. Some minerals tend to cleave along planes at various fixed orientations. Some, like quartz, do not cleave at all, only fracture. Minerals that have cleavage can also fracture along surfaces that are not parallel to their cleavage planes.
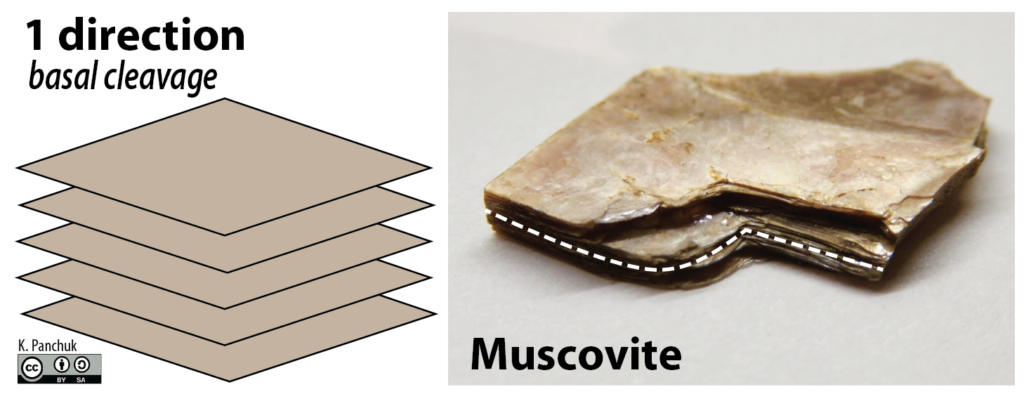
The way minerals break is determined by the arrangement of atoms within them, and more specifically by the orientation of weaknesses within their crystal lattice. Graphite and mica break off in parallel sheets (Figure 5.36).
Other minerals have two directions of cleavage, classified as two directions at 90° (Figure 5.37 top) and two directions not at 90° (Figure 5.37 bottom). While the diagrams of planes on the left of Figure 5.37 make this difference clear, it may be less obvious in practice. The minerals in Figure 5.37 both have two planes of cleavage that are very close to 90°. The white dashed lines mark the edges of the planes, as with Figure 5.36. See if you can find the planes repeated in the images. The images are close-up views of the minerals, only a few cm across. Sometimes you must look very carefully to find cleavage planes.

Some minerals have many directions of cleavage. Figure 5.38 shows minerals with three directions of cleavage. Halite (Figure 5.38 top) has three directions at 90° and calcite (Figure 5.38 bottom) has three directions not at 90°.

There are a few common difficulties that students encounter when learning to recognize and describe cleavage. One is that it might be necessary to look very closely at a sample to see mineral cleavage. The key features in Figure 5.37 are only cm or mm in scale. If crystals are very small, it may not be possible to see cleavage at all. Another issue is that sometimes cleavage is present, but it is poor, meaning the cleavage surface isn’t perfectly flat. Finally it can be difficult to know whether a flat surface on a crystal is a cleavage plane, a crystal face, or simply a surface that happens to be flat. Cleavage planes tend to repeat themselves at different depths throughout the mineral, so if you are unsure whether the surface you are looking at is a cleavage plane, try rotating the mineral in bright light. If cleavage is present, you will generally find that, for a given cleavage direction, all of the cleavage surfaces will glint in the light simultaneously. Crystal faces will also glint in light, but they do not repeat themselves at depth throughout the mineral. The best way to overcome all of these problems is to look at lots of examples. It’s worth it to be able to identify cleavage and fracture, because cleavage is a reliable diagnostic property for most minerals.
Exercise: Can You Pick Out the Broken Piece?
Density
Density is a measure of the mass of a mineral per unit volume, and it is a useful diagnostic tool in some cases. Most common minerals, such as quartz, feldspar, calcite, amphibole, and mica, are of average density (2.6 to 3.0 g/cm3), and it would be difficult to tell them apart on the basis of their density. On the other hand, many of the metallic minerals, such as pyrite, hematite, and magnetite, have densities over 5 g/cm3. If you picked up a sample of one of these minerals, it would feel much heavier compared to a similarly sized sample of a mineral with average density. A limitation of using density as a diagnostic tool is that one cannot assess it in minerals that are a small part of a rock with other minerals in it.
Other properties
Several other properties are useful for identification of some minerals. Some of these are:
- Calcite reacts with dilute acid and will give off bubbles of carbon dioxide.
- Magnetite is strongly magnetic, and some other minerals are weakly magnetic.
- Sphalerite ((Zn,Fe)S) gives off a smell of sulphur when drawn across a streak plate.
- Halite tastes salty.
- Talc feels soapy to the touch.
- Plagioclase feldspar has striations (parallel razor-thin lines etched on the surface) and some varieties show a play of colours when light hits them at the right angle (see the labradorite in Figure 5.28).
Exercise: Are You Ready to Test Your Knowledge of Mineral Properties?
- Note that artificial fingernails may be much harder than natural fingernails. Some materials used for artificial nails are harder than quartz. ↵

