Chapter 6. Relative and Absolute Dating
3 Practice Questions on Absolute Dating
Adapted by Joyce M. McBeth & Sean W. Lacey (2018) University of Saskatchewan from Deline B, Harris R, & Tefend K. (2015) “Laboratory Manual for Introductory Geology”. First Edition. Chapter 1 “Relative and Absolute Dating” by Bradley Deline, CC BY-SA 4.0. View Source. Last edited: 8 Jan 2020
Note that portions of these exercises will require students read through and refer to the “Overview of Relative and Absolute Dating” portion of the lab manual.
6-E2 LAB EXERCISES – RADIOMETRIC DATING
Complete the following chart by calculating the amount of parent isotope remaining for each number of elapsed half-lives. Plot your findings on the graph (Figure 6.11). Make sure you connect the data points on the graph by drawing in the decay curve. Use the completed chart and graph to answer the questions below.
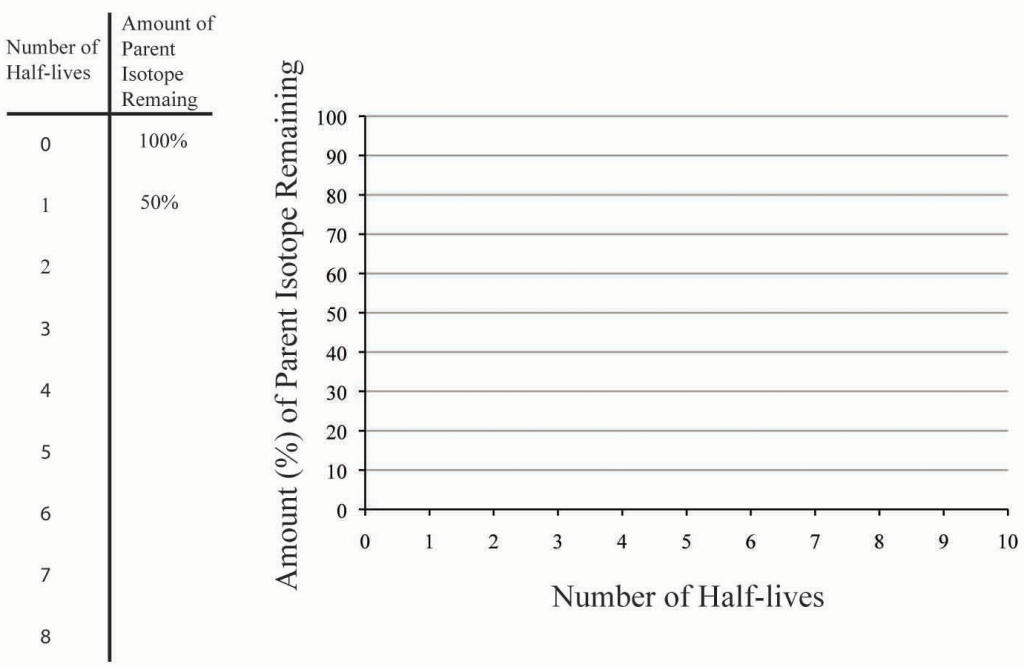
Source: Bradley Deline (2015) CC BY-SA 3.0 view source
10. How much of the parent isotope would be remaining after 7 half-lives have passed?
a. 6.25 %
b. 1.56 %
c. 0.78 %
d. 0.39 %
11. If a radioactive element has a half-life of 425 years, how old would a rock be that only had 3.125 % of the parent isotope remaining?
a. 2125 years
b. 1700 years
c. 2550 years
d. 3400 years
12. Based on your graph above, approximately how much of the parent isotope would be remaining after 3.5 half-lives?
a. 16 %
b. 12 %
c. 4 %
d. 8 %
13. Based on your graph above, approximately how many half-lives have passed when only 35 % of the parent isotope is remaining?
a. 0.75
b. 1.5
c. 2.1
d. 2.5
6.7 LAB EXERCISE – CHOOSING ABSOLUTE DATING METHODS
Determine the most appropriate absolute dating method to determine the ages of the materials in the following questions. Hint: refer to the section on radiometric dating in the overview section of this textbook for half-life values for various radiometric dating methods and examples of how to complete these problems.
14. An Archeologist finds some cotton cloth at a burial site and wants to determine the age of the remains. Which isotopic dating method should they use?
a. Carbon-14
b. Uranium-Lead
c. Potassium-Argon
15. The Archeologist determines there is 16.7 % of the parent isotope remaining in the cloth sample. How old is the burial site? The half-life of carbon-14 is 5730 years.
a. 13,559 years
b. 14,798 years
c. 16,743 years
d. 1.66 billion years
e. 1.81 billion years
f. 2.05 billion years
16. A geologist is trying to date a sequence of sedimentary rocks with abundant fossils. The sedimentary rocks are mostly sandstone. Within the rock sequence there is a distinctive clay layer that under closer inspection is fine-grained volcanic ash. Which of the following is the best way to obtain an absolute date for the sequence of rocks?
a. Carbon date the fossils
b. Potassium-Argon date the sands
c. Uranium-lead date the zircons in the ash
d. Identify the index fossils
17. The geologist determines there is 78.3 % of the parent isotope remaining in the sample that they examine. How old is the sequence of rocks? Hint: your answer to this question will depend on your answer to the previous question!
a. 2022 years
b. 247.8 million years
c. 390.7 million years
d. 2.504 billion years
e. 1.588 billion years
f. 1.202 billion years

